As the global energy demand rises and public favor shifts from carbon-based fuel sources to sustainable energy, research has burgeoned in the area of renewable energy development. Our group approaches this challenge by investigating the relationship between material and energy and the structural level.
Geothermal Lithium Extraction
Hybrid Organic-Inorganic Perovskites
Quantum Dots
Phase Change Materials
Geothermal Lithium Extraction
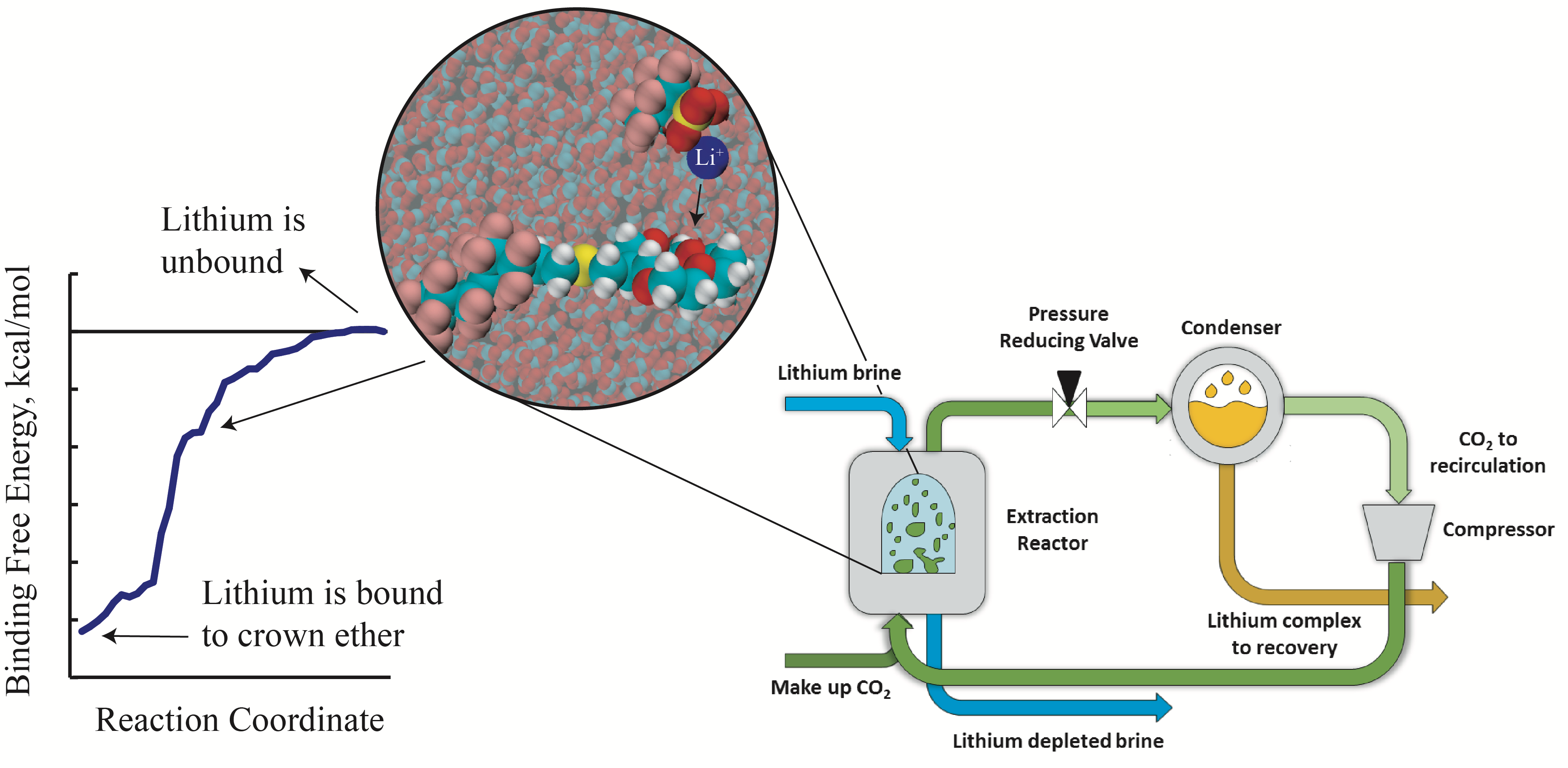
As demand for lithium increases, new avenues for lithium mining must be explored. This, compounded with the shift towards renewable energy, makes lithium extraction from geothermal brine an attractive area of research. In particular, the extraction efficiency and selectivity for various extractants is an important challenge. We use Molecular Dynamics to qunatify these metrics for different extractants and use our molecular insight to predict the structure of potentially favorable extractants.
Researchers: Andrew W. Ruttinger
Related Publications
A. W. Ruttinger, A. Pálsdóttir, J. W. Tester, and P. Clancy, A New Quantitative Metric for the Design of Selective Supercritical CO2 Extractions of Lithium from Geothermal Brine, ChemSusChem (2019).
Hybrid Organic-Inorganic Perovskites (HOIPs)

When processed in solution, Hybrid Organic–Inorganic Perovskite (HOIP)–based solar cells, provide a unique cost advantage over existing solar cell technologies. However, the morphology of the final crystal film is known to be heavily influenced by solvent selection – and these effects remain poorly understood. Studying the nucleation and early stage growth of these materials in solution would provide much needed insight into the effect of solvents on final film morphology. Molecular simulation models and techniques have a significant role to play in this endeavour by uncovering the solvent–solute interactions influencing the formation of moieties that will ultimately nucleate and grow into thin films. Our lab generates models that are capable of accurately predicting the efficacy of certain solvents in the solution processing of lead iodide perovskites. We utilize these models to develop a comprehensive understanding of solvent influence on thin film growth.
Researchers: Blaire A. Sorenson, Oluwaseun “Seun” V. Romiluyi, Spencer Hong
Related Publications
J. M. Stevenson, B. A. Sorenson, V. H. Subramaniam, J. Raiford, P. P. Khlyabich, Y. Loo and P. Clancy, Mayer Bond Order as a Metric of Complexation Effectiveness in Lead Halide Perovskite Solutions, Chem. Mater., 29 (6), 2435-2444 (2017).
B. J. Foley, J. Girard, B. A. Sorenson, A. Z. Chen, J. S. Niezgoda, M. R. Alpert, A. F. Harper, D. Smilgies, P. Clancy, W. A. Saidi and J. J. Choi, Controlling Nucleation, Growth, and Orientation of Metal Halide Perovskite Thin Films with Rationally Selected Additives, J. Mater. Chem. A, 5, 113-123 (2017).
Quantum Dots
Nicknamed artificial atoms, quantum dots have a characteristic length scale in which energy levels are quantized; therefore, altering the size of the quantum dot will control how energy is absorbed or emitted, similar to a true atom. Due to this property, quantum dots are an ideal candidate for use as a photovoltaic material, along with optoelectronic devices. Our research in this area looks at understanding both the mechanism for reaction initiation and the mechanism for quantum dot growth. On an atomic scale, Density Functional Theory allows us to uncover an exquisitely nuanced and informative kinetic mechanism. Then, on a larger molecular scale, Molecular Dynamics provides us insight into the nucleation and growth of smaller quantum dots.
We have extensive experience simulating crystalline quantum dot solids (QDS), from self-assembly to novel ligand choices, to electronic properties. Currently, we are using computational techniques to study the formation of large-scale single crystal QDS, composed of individual colloidal quantum dots connected by epitaxial bridges. The goal is to achieve coherent charge transport through close-to-perfect order. We are using a new reactive force field, SMRFF, that we developed, to model bond formation and uncover the mechanism by which bridges form to drive superstructure creation and the role of defect creation during assembly.
Researchers: Isaiah Chen, Wenxin Qi, Andrew W. Ruttinger, Henry C. Herbol
Related Publications
I. Y. Chen, J. C. daSilva, D. M. Balazs, M. A. Smeaton, L. F. Kourkoutis, T. Hanrath, and P. Clancy, The Role of Dimer Formation in the Nucleation of Superlattice Transformations and Its Impact on Disorder, ACS Nano, 14 (9), 11431–11441 (2020).
J. M. Stevenson,† A. W. Ruttinger,† and P. Clancy, Uncovering the Mechanism Initiating the Nucleation of Lead Sulfide Quantum Dots through a Hines Synthesis,J. Mater. Chem. A., 6, 9402-9410 (2018). [† co-first authors]
B. Lukose and P. Clancy, A Feasibility Study of Unconventional Planar Ligand Spacers in Chalcogenide Nanocrystals, Phys. Chem. Chem. Phys., 13781-13793 (2016).
B. E. Treml, B. Lukose, P. Clancy, D.-M. Smilgies and T. Hanrath, Connecting the Particles in the Box- Creating hexamer nanocrystal clusters within an AB6 binary nanocrystal superlattice, Scientific Reports, 4:6731 (2014).
A. P. Kaushik, B. Lukose and P. Clancy, The role of shape on electronic structure and charge transport in faceted PbX Nanocrystals using Density Functional Theory, ACS Nano, 8(3), 2302-17 (2014).
A. P. Kaushik and P. Clancy, Solvent-Driven Symmetry of self-assembled nanocrystal superlattices – A computational study, J. Comput. Chem., 34, 523-532 (2013).
A. P. Kaushik and P. Clancy, Explicit all-atom modeling of realistically sized ligand-capped nanocrystals, J. Chem. Phys. C, 136, 114702 (2012).
P. Clancy, Nanoparticles: Self-assembly sets its own limits, Nature Nanotech., News and Views, 6, 540-541 (2011).
Phase Change Materials (PCMs)
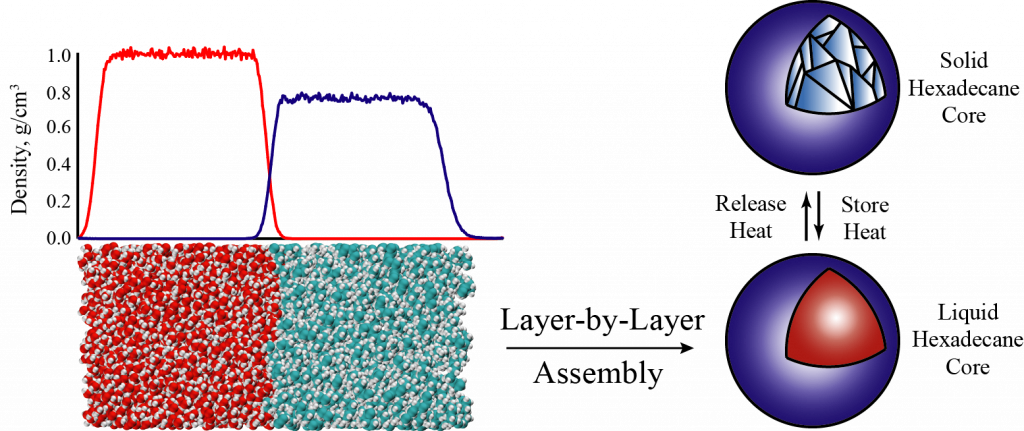
As energy production shifts towards renewables, a growing concern is the need for energy storage media. One particularly attractive class of materials for this challenge are Phase Change Materials (PCMs), which leverage a chemical phase transition for thermal energy storage. This is especially promising for integration into geothermal energy systems because the working fluid is hot and aqueous. To improve stability and thermal conductivity, PCMs can be encapsulated through a process called Layer-by-Layer assembly. Our work in this area dives into investigating the atomic interactions at play during this capsule assembly. Using Molecular Dynamics, we can model the assembly process to quantify the role of different molecular structures to predict potential capsule materials with improved thermal and stability properties.
Researchers: Andrew W. Ruttinger, Xinru Yun


